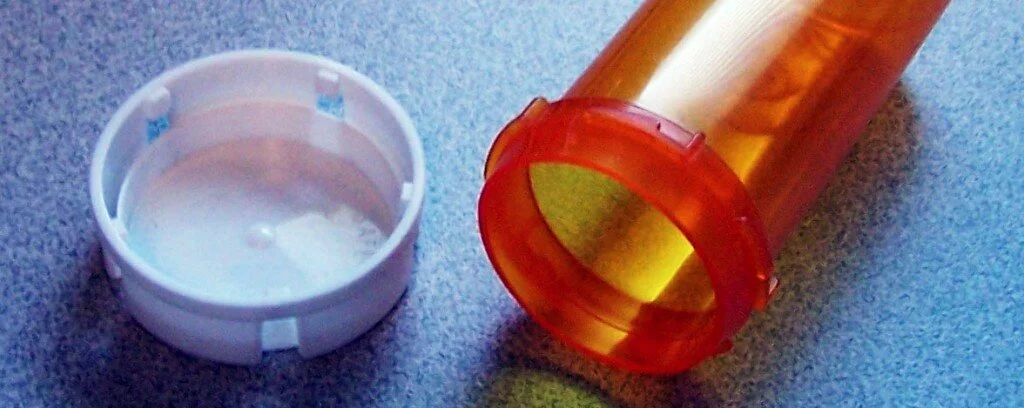
If you have a medication you take regularly, you’re familiar with the problem of waste created by prescription pill bottles. Some people go through several bottles a month — and many municipalities do not recycle this type of plastic. Fortunately, some ultra-creative people have thought up some clever, thrifty ways to re-use these bottles, and keep them out of our landfills, too.
The bottles have a few special characteristics that make them super-useful. For one thing, they are sized uniformly, so they’re perfect for organizational projects, where you want things to line up evenly. Even better, they’re waterproof and air-tight — qualities that you should take advantage of. You might pay a lot for little bottles like these at a place like The Container Store, so just enjoy them for free with your prescriptions instead!
Before you get started, there are two important tips to remember when re-using prescription bottles:
- Wash and dry before re-use, so they are free of any drug residue
- Protect your privacy: soak the bottles to remove the labels
Once you have the bottles clean and label-free, you can begin to see their real beauty and use! Here are some of the top projects, from the kitchen to the garden to the tackle box, that we found around the net:
ORGANIZATIONAL
- The bottles are perfect for holding screws, nails, tacks, and other workshop items. Glue or drill the caps to the underside of a shelf, and let your new storage units “float†underneath
- Long, skinny tubes of makeup such as mascara or lip gloss can be organized to stand up in pill bottles
- The bottles are just the right size to hold a stack of quarters for parking meters or laundromats
- Store fishing lures in the bottles to keep them from tangling with other equipment
GIFTS AND CRAFTS
- Put a few pre-threaded needles, a tiny seam ripper, and some safety pins in a bottle to form an on-the-go sewing kit
- Clean medicine bottles are a great holder for homemade spice mixes such as steak rub or herbes de provence
- Make your own humorous labels for a “funny†medicine and fill with small candies (keep this type of gift away from children to avoid confusing them)
OUTDOORS
- Because they’re water-proof, the bottles make great waterproof matchboxes for camping. Fill them with strike-anywhere matches and glue a striking surface on inside of the lid.
- In the garden, the air-tight bottles work perfectly as seed savers.
- At Halloween, cover white outdoor holiday lights with one bottle each for an amber effect
TRAVEL AIDS
- Use the tiny bottles for a self-serving of salad dressing in your packed lunch
- The bottles are great as travel shampoo and conditioner bottles
CHARITABLE USES
- Some veterinarians will welcome your empty bottles for re-use with pet meds (varies by doctor; inquire with your vet)
- There are some community projects to collect bottles for issuing prescriptions to the homeless
- Similar programs exist for third-world nations… your empties could help with medical aid
What is your favorite use for discarded medicine bottles? We’d love to hear what you’re making!
For more ideas:
- Ways to Use Empty Prescription Medicine Bottles
- What to Do with Old Medicine Bottles
- Uses for Prescription Bottles
- 21 Ways to Recycle Empty Prescription Bottles
 Healthcare reform isn’t just about the public option and paying for doctor’s visits — it’s also about equal, affordable access to life-saving medications for all Americans. That’s why many Big Pharma watchdogs are so disappointed with a recent amendment slipped into healthcare legislation that proposes extending patent protection on biologic drugs, delaying for years the public’s access to affordable follow-on versions.
Healthcare reform isn’t just about the public option and paying for doctor’s visits — it’s also about equal, affordable access to life-saving medications for all Americans. That’s why many Big Pharma watchdogs are so disappointed with a recent amendment slipped into healthcare legislation that proposes extending patent protection on biologic drugs, delaying for years the public’s access to affordable follow-on versions.
What are “biologics� They’re the next big wave in medicine — drugs made not from simple chemical formulations, but from biological components. They’re very expensive, and poised for enormous success:
By 2014, the biggest-selling meds will be biologics, according to an analysis from Evaluate Pharma. Taking the place of Pfizer’s gargantuan drug Lipitor will be Roche’s Avastin, a cancer med expected to account for $9.23 billion in 2014 sales. (Even when you factor in the recent trial disappointments.) The next five top sellers, in order, are expected to be Humira (Abbott Labs), Rituxan (Roche), Enbrel (Wyeth/Amgen), Lantus (Sanofi-Aventis), and Herceptin (also Roche).
Evaluate also predicts that half of the top 100 drugs in 2014 will be biotech meds — a huge change from last year’s level of 28 percent and 11 percent in 2000.
Because biologics are so complex, the system we all know — where patented brand names enjoy a period of exclusivity, then eventually make way to cheaper generics — doesn’t translate perfectly. Biologic “generics†are called “biosimilars,†and they are not seen as generic equivalents. They must be submitted for approvals as new drugs and do their own clinical trials, etc.Â
The Eshoo-Barton amendment, named for sponsoring Representatives Anna Eshoo (D – Calif.) and Joe Barton (R – Texas), would give brand-name biologic drugmakers 12 years of market exclusivity. By comparison, President Obama favors seven years, and Rep. Henry Waxman (D –Calif.) feels that the public should have access to “generic†biologics after just five years. By contrast, says Medical News Today, “The Biotechnology Industry Organization maintains that there should be a minimum 14 years of exclusivity to account for a development process that on average takes 10 years and $1.2 billion for a product to reach market.â€
5, 7, 12, or 14 years? As you can see, there is a real difference of opinion on this subject. One person who has written extensively on this is author James Love on the Huffington Post. Here he explains why this amendment is harmful:
The Eshoo/Barton amendment, which has the support of many newly pro-PhRMA democrats, will extend the period of monopolies for biologic medicines, when compared to the original Waxman text. The only question is how long. Part of the harm will be the longer period prohibiting generic suppliers from relying upon evidence that medicines are safe and provide therapeutic benefits. Much of the other harm will come from a number of technical changes in the bill that make it much easier for incumbent firms to block entry through technical issues, extended litigation, and ever-greening of protection from small medically unimportant changes in protected medicines.
This is essentially a case of innovation versus access. Drug companies want protection from the risks and costs borne in the creation and testing of new drugs; patient advocates say that Big Pharma (or Big Biotech, if you like) already make large profits and that the public deserves access to affordable biosimilars in a more timely fashion. “Entities that support longer periods of exclusivity — such as universities, biotech companies and venture capitalists — are ‘fighting to protect inventors’ rights and ensure more thorough clinical trials.’ On the other side, consumer groups, labor unions, insurers and generic drug manufacturers ‘see shorter exclusivity as the way to deliver safe, affordable and quality drugs to patients and open the marketplace to increased competition,’†explains Medical News Today.
The latest high-emotion development is blogger Jane Hamsher’s “Are You Or Someone You Know Paying $50,000 A Year For Drugs?†It paints an ugly picture of what happens to people who cannot affording life-saving biologics. A few days later, Rep. Eshoo responded to this and other online attention with a blog post on The Hill’s Congress Blog titled “Setting the record straight on our health care legislation.†If you check in with these two articles, you’ll have the latest from both side of the “biologic generics” debate.
Our mission, as always, at EDrugSearch.com is to improve the American public’s access to safe, quality medications at an affordable cost.
For more information:
- Should We Be OK With The PhRMA Deal With White House?
- US House Panel Backs Exclusivity for Biologic Drugs
- Pay or die: Deadly Pharma amendment in HCR going right under radar
Save money on popular meds as they go generic in 2010

We’re nearing the end of the year, and many people are already in open enrollment for next year’s health coverage, which means it is a great time to plan ahead for your 2010 drug expenditures. No, you can’t know every medication you are going to need, but if you are on a popular, patented maintenance drug such as Lipitor or Cozaar, you’ll be pleased to hear that their patents are expiring in 2010, opening the field for more-affordable U.S. generic versions.
In the meantime, as you do your research, you will see that many are already available as generics, manufactured outside of the U.S. There is usually a considerable cost savings.
Here are all the details of some of the highest-profile drugs that have expiring patents as of the end of 2009, or during 2010 — and how and where to get the generic versions.
ARICEPT (donepezil). Pfizer makes the acetylcholinesterase inhibitor Aricept for the treatment of Alzheimer’s disease. Aricept is scheduled to lose patent protection in the U.S. in 2010, but generic versions are already available.
COZAAR (losartan). Merck’s Cozaar is an angiotensin II receptor antagonist, used to treat hypertension. Cozaar’s patent protection in the U.S. will expire in 2010. In the meantime, generic losartan is available here.
LEVAQUIN (levofloxacin). Johnson & Johnson’s antibiotic drug Levaquin will outlive its patent protection in 2010. However, until that time, you can purchase generic levofloxacin here.
LIPITOR (atorvastatin). Pfizer has the best-selling drug in the world in Lipitor, which treats high cholesterol. In the US, the basic patent for Lipitor expires in March 2010, while the second patent covering the calcium salt of atorvastatin expires in June 2011. It is not yet clear exactly which date will see widespread availability of generic Lipitor in the U.S., but it is coming. In the meantime, generic atorvastatin is available here.
XENICAL (orlistat). Roche’s Xenical is an obesity treatment that prevents the digestive system from absorbing fats. Its patent expires in December of this year. (Note: In some countries, including the U.S., orlistat is available over-the-counter as GlaxoSmithKline’s Alli.)
VALTREX (valacyclovir). GlaxoSmithKline’s Valtrex is a treatment for herpes simplex and herpes zoster (shingles). Valacyclovir was recently approved for generic production, and so generic versions will be on the U.S. market by the end of 2009. As of this writing, generic valacyclovir is available here.
If you’re interested in the business ramifications for the pharmaceutical companies, CNN ran a good article on the topic, and so did Chemistry World.
Enjoy using EDrugSearch.com to knowledgeably plan your medication choices for the coming year.
As the 2009/2010 influenza season progresses, there is cause for worry. Flu activity is already widespread in 46 states, according to the Center for Disease Control (CDC), and the 2009 novel H1N1 flu virus, popularly known as the “swine flu,†is on the rise. Meanwhile, the swine flu vaccine is difficult to obtain, leaving many unprotected from this new strain. Fortunately, the antiviral treatments Tamiflu and Relenza are still largely effective against the swine flu.
Why is there so much worry about pandemic flu this year? Regular flu kills 36,000 people in the US each year, but the swine flu variation has the potential to kill many more. The latest update from the CDC shows that swine flu has caused at least 95 children’s deaths since April. Why is this strain so dangerous?
- It is a new combination (made of human, swine, and avian elements) and virtually no one has natural immunity to it
- The vaccine is not yet widely available
- This strain attacks the young and healthy population in disproportionate numbers
There is some hopeful news from the CDC in terms of protecting yourself:
Almost all of the influenza viruses identified so far are 2009 H1N1 influenza A viruses. These viruses remain similar to the virus chosen for the 2009 H1N1 vaccine, and remain susceptible to the antiviral drugs oseltamivir and zanamivir with rare exception.
Influenza antiviral drugs are prescription drugs (pills, liquid, or inhaler) that decrease the ability of flu viruses to reproduce. It is advised that you begin them within two days of getting influenza. Many people ask about taking anti-viral drugs before they show symptoms, but according to the CDC, pre-exposure antiviral chemoprophylaxis should only be used in limited circumstances, and in consultation with your doctor.
The drug oseltamivir (trade name Tamiflu, from Roche) is one of two drugs that is approved for treatment of swine flu. It is taken orally, in a pill form or a suspension. Common side effects include: nausea, vomiting, diarrhea, abdominal pain, and headache. Less frequently, delirium, hallucinations, rash, hepatitis, and allergic reactions including anaphylaxis can occur. There is some concern that oseltamivir can cause the desire to self-harm, and this seems more pronounced in children. People with the flu, particularly children, may be at an increased risk of self-injury and confusion shortly after taking Tamiflu and should be closely monitored for signs of unusual behavior. However, oseltamivir is approved by the FDA for use in children 1 year of age or older. In addition, in July 2009, the FDA issued an Emergency Use Authorization for the use of Tamiflu in pediatric patients younger than 1 year of age.
The drug zanamivir (trade name Relenza, from GlaxoSmithKline) is the other medication approved to treat swine flu. It is an inhaled powder that comes with a disk inhaler. It can cause side effects, especially in those with asthma or other chronic lung disease. Decreased respiratory function and bronchospasm have been reported with use of zanamivir. Zanamivir is generally not recommended for use in persons with underlying lung disease such as asthma and chronic obstructive pulmonary disease. Other side effects reported by less than 5% of those who have used this drug are diarrhea, nausea, sinusitis, nasal infections, bronchitis, cough, headache, and dizziness. Relenza is approved for treatment in children 7 years and older, but only those without underlying respiratory or heart disease, including asthma.
One final caution as you navigate flu season is to beware of some unscrupulous online vendors selling fake flu drugs to capitalize on the public’s fear. The Food and Drug Administration recently issued a warning to consumers to be careful not to purchase counterfeit medications. You may rest assured that eDrugSearch.com deals only with reputable, licensed pharmaceutical suppliers in the U.S. and Canada. Our advanced search features enable members to identify pharmacies with specific licensing requirements, third-party accreditations, Better Business Bureau memberships, and more. We offer a feedback system so consumers can share their ratings of the pharmacies they deal with. Browse the ratings, and enjoy peace of mind as you choose among our consumer-tested pharmacy partners for your flu season needs.
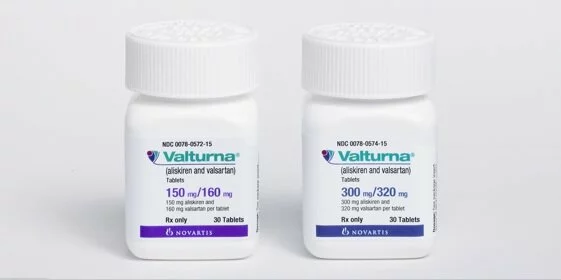
A new blood-pressure treatment has come on the market that is of potential interest to hypertension patients whose blood pressure is not being adequately controlled with monotherapy.
Last month, the US Food and Drug Administration (FDA) approved Novartis’ Valturna (aliskiren and valsartan), which combines in a single pill valsartan, the active ingredient in Diovan — the top-selling branded high blood pressure medicine — and aliskiren, the active ingredient in Tekturna — the only approved direct renin inhibitor (DRI).
Valturna is the first and only medicine to target two key points within the renin system, an important regulator of blood pressure. It is suitable for patients whose high blood pressure has not been adequately controlled on aliskiren alone, or by an angiotensin receptor blocker, such as Diovan, alone. It could also be considered as an initial treatment for patients who are likely to need multiple drugs to achieve their blood pressure goals.
Valturna targets the renin angiotensin aldosterone system (RAAS) in two ways. The valsartan component blocks, at the receptor level, the action of angiotensin II, which causes blood vessels to tighten and narrow. The aliskiren component directly inhibits renin, an enzyme produced by the kidneys that starts a process that leads to formation of angiotensin II. This helps blood vessels relax and open.
Because Valturna combines two different types of blood pressure drug, it offers greater blood pressure reduction than either drug would alone. It also seems conceivable having both medications available in one pill would help consumers with drug costs and insurance coverage.
Cautions: Women of child-bearing age should be particularly careful with this medicine. AVOID USE IN PREGNANCY. Do not take if you are breastfeeding or if you are planning to become pregnant. If pregnancy is detected, discontinue Valturna as soon as possible. This is because drugs that act on the renin system can cause injury and death to the unborn baby. Other serious reactions to be aware of when taking Valturna are a risk of head and neck angioedema (a rapid swelling of the flesh), and hypotension (low blood pressure). If either of these are suspected, call your physican immediately. Side effects of Valturna are the same as those of its two component medications, valsartan and aliskiren. Patients may experience tiredness, sore throat, runny nose, diarrhea, upper respiratory tract infection, urinary tract infection, flu, or dizziness. Consult your doctor before taking this or any other medication.
A new paper in the scholarly journal Trends in Ecology and Evolution suggests that oral contraceptives may influence women’s choice of mates, with potentially enormous effects on their quality of life and their ultimate reproductive success.
Commonly used hormonal birth control methods such as Yasmin, Seasonale, Ortho Tri Cyclen, and Loestrin, while extremely effective at preventing unwanted pregnancy, may well have previously unsuspected effects on a woman’s choices of both long-term and short-term partners. This is of interest to the more than 100 million women around the globe who use oral contraceptives, and their sexual partners, too.
The paper, published October 9th, was authored by biologists Dr. Alexandra Alvergne and Dr. Virpi Lummaa at the University of Sheffield in England. It analyzed a large body of research that had gone before. Writes Dr. Alvergne, lead author of the study, “The use of the pill by women, by changing her mate preferences, might induce women to mate with otherwise less-preferred partners, which might have important consequences for mate choice and reproductive outcomes.”
Researchers have long known that women make different sexual choices during their most fertile phase than they do during the rest of the month. This is called “cycling mate preferences,†and quite simply, it means that most women choose more masculine and symmetrical faces when they are ovulating or close to ovulation. They are also uncharacteristically attracted to dominant males, and most likely to engage in infidelity at this time in their cycle. During the rest of their cycle — periods when the women are more likely to be infertile — they are more attracted to less masculine faces, and men who are better choices as long-term partners.
Oral contraceptives work by mimicking pregnancy and stifling ovulation, which flattens out the hormonal cycles usually experienced by a fertile woman. Why is this a concern? Says the paper:
New evidence suggests that the pill, by eliminating oestrus, changes the natural cycling preferences in women for markers of both genetic quality and compatibility in mates as well as natural cycling attractiveness to men. As compared with normally cycling women, pill users show no or weaker preferences for facial and vocal masculinity. For instance, the preferred face shape is more masculine during the high conception-probability phase of the menstrual cycle in non-pill users, but pill users do not show similar preference.
The possibility is that a woman could make a choice influenced by birth control that is perhaps not in her best reproductive interests. Even worse, with our cultural customs of lifelong mating and marriage, a woman could change her contraceptive practices and find that she is not attracted to her mate in the same fashion as she was when she was under the influence of prescribed hormones. Either of these outcomes could be devastating to a paired couple.
Oral contraceptives are still regarded as one of most effective forms of birth control available to women (they trail the intra-uterine device by a fraction of a percentage point, but the IUD is appropriate for fewer patients, and is rarely prescribed for women who have not already gone through childbirth). Birth control pills are affordable, simple to obtain, and work extremely well. However, this paper has given us all something to think about: the possibility that altering the hormonal cycles of the human female en masse could have significant reproductive effects for individual women, and the entire species, too.
More research is forthcoming in this arena, and the results are sure to be fascinating.
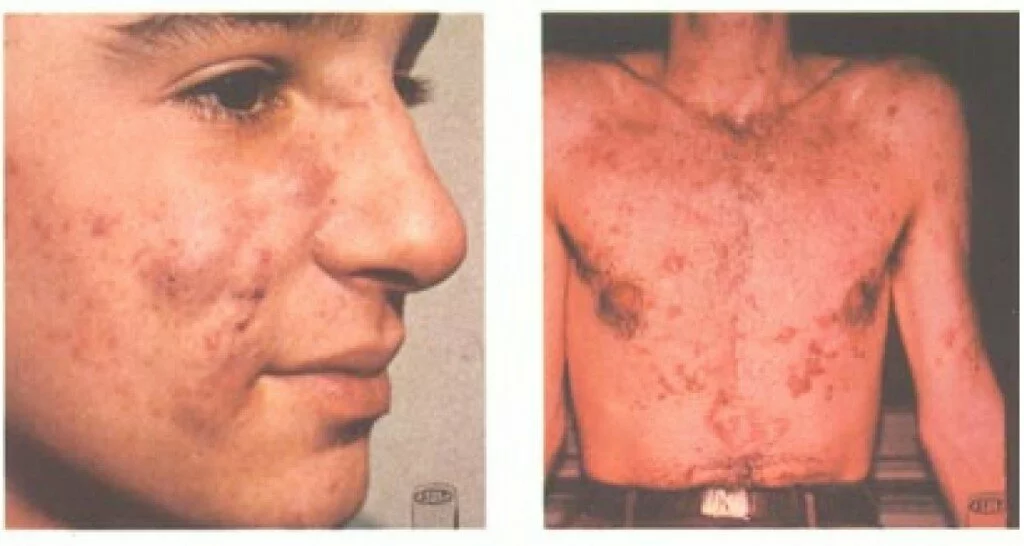 Millions of people suffer from severe, debilitating acne that does not respond to over-the-counter treatments such as acne washes and benzoyl peroxide, and even resists prescription antibiotics. Ongoing severe acne can cause physical effects such as pain, infection, and scarring, and social and emotional effects such as shame and low self-esteem. According to the Acne Resource Center:
Millions of people suffer from severe, debilitating acne that does not respond to over-the-counter treatments such as acne washes and benzoyl peroxide, and even resists prescription antibiotics. Ongoing severe acne can cause physical effects such as pain, infection, and scarring, and social and emotional effects such as shame and low self-esteem. According to the Acne Resource Center:
Of the 85% of teenagers (between the ages of 12 and 24) that suffer from acne, 25% will have permanent scars ranging from severe to light. The American Dermatologist Association finds that:
- 20% of all adults have active acne
- 60 million Americans have active acne
- 20 million Americans have acne badly enough to cause scars.
Acne affects people regardless of age, gender or race. While thoroughly treatable, of those who suffer from acne:
- 11% will see a physician
- 20% will go to a skin care center
- 30% will use an over-the-counter medication from a drug store or pharmacy
- 40+% will do nothing
The concept of 40% of sufferers doing nothing is tragic. Since 1982, we have had access to a prescription drug that treats serious cases of cystic or nodular acne. That was the year isotretinoin, under the trade name Accutane, was approved by the FDA for the treatment of severe acne. Its exact mechanism is not known, but it is thought to limit the production of sebum. Most patients see significant improvement, or remission, after a 15-to-20-week course of treatment with isotretinoin.
The drug was controversial from the start, as it is known to cause teratogenic birth defects. All physicians and patients involved with this treatment must abide by a strict pregnancy risk-management program. Isotretinoin has some other serious side effects, too. Mild side effects involve skin dryness and eye dryness, rashes, headaches, hair thinning, and backaches. More serious side effects include inflammatory bowel disease, alopecia, catacracts, blood glucose problems, and more. Because of these side effects, it is only recommended to treat the most severe cases of disfiguring acne, and patients must pay close attention to side effects.
Availability of the drug has changed this year, resulting in some confusion in consumers. Earlier this year, in July 2009, Swiss drugmaker Roche announced that it was discontinuing its sales of Accutane and moving to delist with the FDA. This was not done for safety and efficacy reasons (although Roche has had to defend itself in many lawsuits), but rather because generics of Accutane (isotretinoin) have been available since 2002 and now dominate the market. Accutane itself had only held on to a 4-5% share of market, and it did not make business sense Roche to keep selling it.
Accutane is still available in many countries outside of the United States. Within the United States, its generic form, isotretinoin, is available, and this drug is also marketed as under several different names within the United States: Claravis, Amnesteem, Sotret are a few of the best-known.
Accutane being retired in the U.S. has caused a great deal of confusion among consumers. Posts on many popular acne-related message boards reveal people trying to compare side effects and benefits of several different brand names that are all essentially isotretinoin. To clarify: there is no difference between Accutane, Sotret, Claravis, or generic isotretinoin except for their makers and packaging.
If your doctor has prescribed a cream instead of a pill, topical isotretinoin is marketed under the trade name Isotrex.
If you are one of the millions suffering from acne, know that you do have options, and your condition can improve.
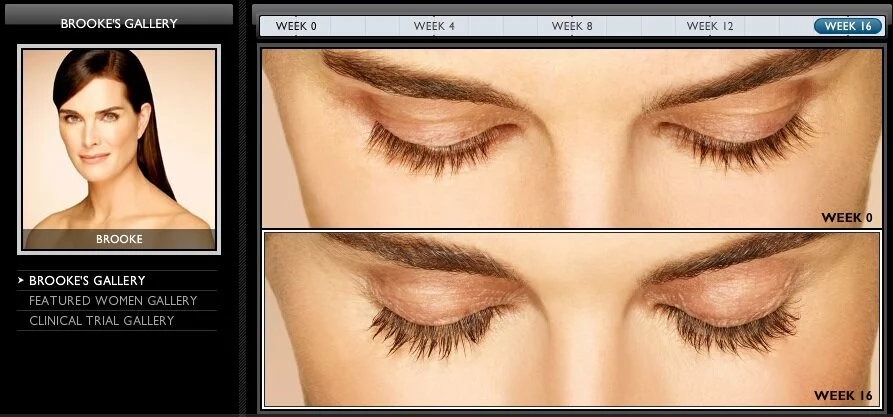
Some people have very sparse eyelashes, and this can cause functional problems with the eye as well as anxiety about one’s appearance. Fortunately for people suffering from ” eyelash hypotrichosis,” a brand-new treatment was approved by the FDA in December 2008. The product is called Latisse, it has sultry-eyed Brooke Shields as a spokesmodel, and it is a 0.03% solution of bimatoprost marketed with an applicator brush.
Latisse is meant to be dabbed along the base of the upper lash line (the lower is too sensitive), causing hair there to grow in much more thickly and darkly than before. Its effects are not permanent, and if it is discontinued, the lashes will return to their original condition within weeks or months.
Technically, bimatoprost is a prostaglandin analog that affects eye pressure. As a generic, and under the trade name Lumigan, the solution is used to control the progression of glaucoma and manage ocular hypertension. Although the main ingredient is the same, using bimatoprost or Lumigan, which are serious ocular pressure meds, to lengthen or grow eyelashes is an unapproved, off-label use. (In fact, eyelash growth was a surprise side effect of these meds.) Some are tempted to swap these pharmaceuticals freely, but doctors warn that it can be harmful to your health profile and insurance history to be have a history of ordering glaucoma drugs when you are merely trying to treat a cosmetic problem.
Commercials for Latisse have been in heavy rotation on television and in glossy women’s magazines lately. If you aren’t paying close attention, you might think you are looking at a mascara ad, but mentions of the treatment being the “first and only†treatment approved by the FDA to treat hypotrichosis make it clear that this is not just make-up. The glam may have gone a little too far, though, says the FDA. MSNBC reports:
Last week, Latisse made headlines when the FDA sent a warning letter to Allergan stating the promotional materials posted on the product’s Web site were “misleading because they omit and minimize risks associated with Latisse.†Among the risks, the FDA notes, is that the active ingredient can cause hair to grow in other places besides the lash area, cause inflammation of the cornea — and can make lighter-colored eyes turn brown.
Meanwhile, an article at BNET says the FDA also accused Allergan of downplaying infection risks.
We went directly to the Latisse website to get their own description of the side effects. Here’s a summary: About 4% of users experience itchy and/or red eyes. Less common side effects include skin darkening, eye irritation, dryness of the eyes, and redness of the eyelids. It is very important not to use Latisse directly on the eye, or on the lower eyelid. It should only be applied to the UPPER eyelid with a sterile applicator. Latisse may cause darkening of the eyelid skin which may be reversible, and it may also cause increased brown pigmentation of the colored part of the eye which is likely to be permanent.
The information is there, but not particularly prominent. Changing the color of the eye is a huge side effect, and we’re curious how the public will react. Latisse is a very popular product, getting huge traffic on beauty boards as people compare notes on how to solve their lash problems without makeup. It will be interesting to see what develops as the FDA and Allergan come to an agreement on how to present side-effect information fairly.

Earlier this month, a closely-watched trial over the osteoporosis drug Fosamax ended in mistrial, to the frustration of nearly everyone involved. The trial was marked by great tension, with a deadlocked jury, reports of threats of physical violence, and a judge-ordered cooling-down period.
What could cause such intense drama? Well, this was just one of approximately 900 state and federal cases pending against Fosamax, alleging that that medication causes osteonecrosis of the jaw (the death of jawbone tissue). In large part, the tension in the Manhattan courtroom was that this trial — the first — was supposed to be an indicator of how these hundreds of similar cases might proceed. The other major factor is that it is notoriously difficult to “prove†drug-related injuries, and this difficulty was definitely shown in the frustration and tension among jury members.
Millions of women have taken Fosamax (alendronate), a Merck drug that was approved in 1995 to treat osteoporosis associated with menopause, and in 1997 to prevent osteoporosis itself. Until the recent introduction of some competing medications, it was one of the most popular drugs in the U.S. It is still prescribed millions of times per year to women suffering from bone loss.
In short, Merck’s defense on this topic is that there is no definitive evidence that Fosamax causes the death of jaw tissue, while plaintiffs and their lawyers insist that Merck overpromoted Fosamax without warning doctors about the potential for jaw injury. Obviously, no conclusions were reached.
Of course, this is of great interest to women who may have taken, or are considering taking, Fosamax. Only your doctor can decide what is the right choice for you, but we thought we would enumerate the other medications available for treating osteoporosis for those doing research on Fosamax alternatives. One very important thing to be aware of it is that Fosamax is not the only osteoporosis drug in this class (the bisphosphonates), which all have a similar mode of action. Fosamax is being talked about most in the media, but all are associated with some amount of risk of damaging the jaw.
If you do choose to take a bisphophonate, know these facts:
- A high proportion of jaw injuries occur following high-dose intravenous administration, so that is a particularly risky way to take the medication.
- As many as 60% of the cases are preceded by a dental surgical procedure involving the jaw. In short, women who are expecting major dental work should consider delaying treatment with Fosamax, Boniva, Actonel or similar drugs until after their dental surgery.
Here are the main alternatives on the market for Fosamax:
- Actonel (risedronate) is a bisphosphonate manufactured and marketed by Procter & Gamble and Sanofi-Aventis. It belongs to the same family of drugs as Fosamax, and may be associated with the same jaw side effects.
- Boniva (ibandronate) is also a bisphosphonate, manufactured and marketed by GlaxoSmithKline and Roche Laboratories. It is a competitor to Fosamax, and may also affect the jaw tissue.
- Evista (raloxifene) is NOT a bisphosphonate , but rather an oral selective estrogen receptor modulator from Eli Lilly and Company that affects bones through estrogen. Evista may be an interesting alternative for women concerned about their jaw health or planning oral surgery.
Also this month, The Wall Street Journal published an article called “From the Osteoporosis Front, Updates on Potential New Drugs.†These are the up-and-comers in clinical trials and going before the FDA. The story covers the latest news about Wyeth’s Viviant, Pfizer’s Fablyn, Amgen’s denosumab, and other upcoming treatments for osteoporosis.
If you are interested in following the Fosamax case that ended in a mistrial, it is expected to be re-tried in the spring. The name of the case is “In re Fosamax Products Liability Litigation, U.S. District Court, Southern District of New York (Manhattan), No. 06-1789.”

Erectile Dysfunction drugs such as Viagra, Cialis, and Levitra have made huge strides in treating male sexual dysfunction — but have women been left behind? At this writing (September 2009), no treatment has been approved by the FDA for Female Sexual Dysfunctions (FSD), which are estimated by the Journal of the American Medical Association to affect 43% of American women (about 40 million) at some point in their lives.
It’s not for lack of trying. Pharmaceutical makers would love to tap into a market that’s the other 50% of the population. They are assertively researching the problem. So why has a result not come forward?
One issue is that, with men, “sexual dysfunction,†no matter whether the cause is mental or physical, is likely to respond as a pretty straightforward plumbing problem: Viagra and its counterparts, the Phosphodiesterase Type 5 Inhibitors, are vasodilators, meaning that they open up blood flow to the area, allowing an erection to be achieved and retained more easily. (More information on the effects of these vasodilators on women is below.)
But women’s sexuality is more complex and less understood, often caused by a range of physical, emotional, and psychological conditions. Simple manipulation of bloodflow only goes so far. Another element is that women have unique hormone issues, which often cause low libido, as they age. Finally, some researchers are experimenting with methods that place sexuality in the mind.
The good news is that research IS progressing. Here’s an update on where the various methods are:
THE BIG THREE (Viagra, Cialis, Levitra)
Researchers are looking into using male ED drugs on women suffering from FSD. In April of this year, a study by the American Physiological Society was described that compared the effects of three drugs used for erectile dysfunction (Viagra (sildenafil), Levitra (vardenafil), and Cialis (tadalafil)) on female rats. The researchers discovered that these drugs significantly increased blood flow to female sexual organs, and show promise for treating similar conditions in human women. Another study has shown that Viagra can help with orgasms in women who are taking SSRIs and suffering sexual dysfunction as a side effect.
There is promise here, but it is all forward-looking. The FDA has not approved these medications for use by women. We will be closely following the progress of this research.
HORMONAL TREATMENTS
The hormone testosterone drives sexual desire, and as women age, they typically lose some of both. There are some doctors that prescribe testosterone off-label to women to help with low desire, but this process is not approved by the FDA. There have been flops along the way to a hormonal treatment. Intrinsa, a testosterone patch for women, was turned down by the FDA in 2004 due to worries about long-term safety, and PT-141 was a nasal spray that was being tested to raise desire in both men and women when it was quashed by the FDA for having possible blood-pressure-related side effects.
In upcoming news, though, BioSante Pharmaceuticals of Illinois is in late-stage studies of a testosterone gel intended to remedy libido loss after menopause. The product, called LibiGel, is smeared on the upper arm and promotes a long, slow build-up of testosterone in women. We’ll be eagerly watching to see if LibiGel is approved by the FDA.
NEUROTRANSMITTER ADJUSTMENTS
A nonhormonal treatment for FSD is in late-stage testing. Boehringer Ingelheim’s Flibanserin, a pill to be taken daily, acts on serotonin receptors in the brain. Early reports suggest that the medication was not useful for treating depression (its intended use), but had the curious side effect of increasing desire in females. The company is now researching the drug’s effects directly in younger, premenopausal women who are suffering from low desire.
The reality is that there is no approved pharmaceutical treatment on the market for a condition that troubles millions of women. The good news is that there are a few promising treatments on the horizon, and we encourage women to follow issues like this in the news so that they will know when relief becomes available. Some of the companies mentioned in this article are conducting clinical trials of these medications, so if you are deeply interested and want to participate, that is a possibility, too. We should see some major changes in the field in the next few years.
-
-
-

-
How to Safely Buy Prescription Drugs Online from Cary Byrd on Vimeo.
-
Search Blog Posts
-
Blogroll
- Boerne Church
- Boerne website design
- Bullet Wisdom
- Bulverde Church
- Christian Social Network
- DrugWonks.com
- Eye on FDA
- Health 2.0
- Kevin, M.D.
- Pharm Aid
- Pharma Marketing
- PharmaGossip
- Pharmalot
- River Crossing Homes for Sale
- San Antonio Asphalt
- San Antonio Life Insurance
- San Antonio Pressure Washing
- The Health Care Blog
- The Peter Rost Blog
- Watch Christian Videos
- World Vision
-
Our Healthcare100 Ranking
-
Trending Content
-
Archives
-
Tags
adverse drug reactions big pharma Canadian drugs canadian pharmacies canadian pharmacy consumer reports Dave Ramsey discount drugs divine healing Drug costs drug prices Drug reimportation eDrugSearch.com FDA Fosamax healing scriptures Health 2.0 health blog directory health blogs healthcare100 healthcare reform Hypertension Jehova Rophe Jesus Christ Lipitor Medi Share miracles nabp online pharmacy dictionary osteoporosis Pharmacies pharmacy spam phrma Prescription drugs promote health blog Proverbs 3:5-8 reimportation relenza Roche saving money submit health blog Tamiflu The Great Physician The Lord our Healer Total Money Makover Live -
